 I have deliberately constructed the word entropy to resemble as much as possible the word energy, since both quantities to be named by these words are so closely related in their physical meaning that a certain similarity in their names seems appropriate to me. As I deem it better to derive the names of such quantities - that are so important for science - from the antique languages, so that they can be used without modification in all modern languages, I propose to call the quantity S the entropy of the body, after the Greek word for transformation, ἡ τροπή. Translation: Searching for a descriptive name for S, one could - like it is said of the quantity U that it is the heat and work content of the body - say of the quantity S that it is the transformation content of the body. 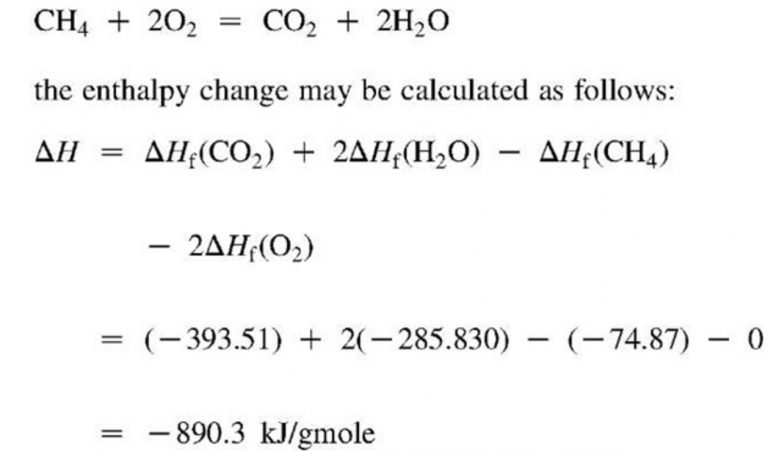
Williams, Richard (2009), "The Sackur–Tetrode Equation: How entropy met quantum mechanics", APS News, 18 (8).(This derives a Sackur–Tetrode equation in a different way, also based on information.) (2013), "Physics of uncertainty, the Gibbs paradox and indistinguishable particles", Studies in History and Philosophy of Science Part B, 44 (4): 480–489, Bibcode: 2013SHPMP.44.480K, doi: 10.1016/j.shpsb.2013.08.007. (2002), Logic of Thermostatistical Physics, Springer-Verlag, Chapter 3: Kinetic theory of gases. ^ Ben-Naim, Arieh (2008), A Farewell to Entropy: Statistical Thermodynamics Based on Information, World Scientific, ISBN 978-981-270-706-2, retrieved.The NIST Reference on Constants, Units, and Uncertainty. ^ "2018 CODATA Value: Sackur–Tetrode constant".^ "2018 CODATA Value: atomic mass constant"."100th anniversary of the Sackur–Tetrode equation". 
elementaren Wirkungsquantums" (The universal significance of the so-called elementary quantum of action), Annalen der Physik 40: 67–86. Sackur (1913) "Die universelle Bedeutung des sog. Nernst zu seinem 25jährigen Doktorjubiläum gewidmet von seinen Schülern (Halle an der Saale, Germany: Wilhelm Knapp, 1912), pages 405–423. Sackur, "Die Bedeutung des elementaren Wirkungsquantums für die Gastheorie und die Berechnung der chemischen Konstanten" (The significance of the elementary quantum of action to gas theory and the calculation of the chemical constant), Festschrift W. Sackur (1911) "Die Anwendung der kinetischen Theorie der Gase auf chemische Probleme" (The application of the kinetic theory of gases to chemical problems), Annalen der Physik, 36: 958–980. ^ Sackur published his findings in the following series of papers:.  
Tetrode (1912) "Berichtigung zu meiner Arbeit: "Die chemische Konstante der Gase und das elementare Wirkungsquantum" " (Correction to my work: "The chemical constant of gases and the elementary quantum of action"), Annalen der Physik 39: 255–256. Tetrode (1912) "Die chemische Konstante der Gase und das elementare Wirkungsquantum" (The chemical constant of gases and the elementary quantum of action), Annalen der Physik 38: 434–442. (1999), An Introduction to Thermal Physics, Addison Wesley Longman, ISBN 7-7 The individual entropy terms will not be absolute, but will rather depend upon the standards chosen, and will differ with different standards by an additive constant. If each logarithmic argument were divided by an unspecified standard value expressed in terms of an unspecified standard mass, length and time, these standard values would cancel in the final result, yielding the same conclusion. Strictly speaking, the use of dimensioned arguments to the logarithms is incorrect, however their use is a "shortcut" made for simplicity. The Sackur–Tetrode equation expresses the entropy S. It is named for Hugo Martin Tetrode (1895–1931) and Otto Sackur (1880–1914), who developed it independently as a solution of Boltzmann's gas statistics and entropy equations, at about the same time in 1912. The Sackur–Tetrode equation is an expression for the entropy of a monatomic ideal gas. Expression of monatomic ideal gas entropy 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
